This website is under construction. Please visit after a week for the complete website experience.
Paediatric Surgery, Shaikh Zayed Hospital, Lahore

Vascular Malformations
Table of Contents
-
Introduction
-
Capillary Malformation
-
Lymphatic Malformation
-
Venous Malformation
-
Arteriovenous Malformation
-
Mixed Syndromes
Introduction
Vascular malformations arise due to diffuse or localized errors of development. Any segment of the vascular tree may be affected including capillary, lymphatic, venous and lymphatic channels. Distinct from vascular tumors, which exhibit postnatal growth due to endothelial proliferation, vascular malformations tend to exhibit growth proportional with that of the child. They are divided into:
Capillary Malformations
Lymphatic Malformations
Venous Malformations
Arteriovenous Malformations
Mixed Syndromes
Capillary Malformations
Correct name for the port wine stain. Capillary malformations present at birth usually as sporadic, singular, flat, pink red, cutaneous patches. There is a propensity for the head and neck but they can be found anywhere on the body. The malformations can be localized or extensive. Occasionally, capillary malformations are associated with hypertrophy of the soft tissue, muscle and bone. It can cause limb length discrepency. Overtime can darken and cause a knobby appearance.
The presence of a capillary malformation (CM) in certain patterns may signal an underlying structural abnormality. A CM overlying the cervical or lumbar spine may be associated with an occult spinal dysraphism or tethered cord. An encephalocele or ectopic meninges may be beneath a CM of the occiput. Capillary malformations in the distribution of the ophthalmic and maxillary branches of the trigeminal dermatomes warrant evaluation for Sturge-Weber syndrome. The syndrome consists of a facial capillary malformation with ipisilateral ocular and leptomeningeal vascular anomalies. Extensive involvement of the leptomeninges manifests as seizures, contralateral hemiplegia and variable motor and cognitive delays.
Treatment
Most capillary malformations do not require intervention. The treatment for capillary malformations is focused on lightening the stain. Pulsed dye laser treatment is the preferred treatment. Capillary malformations of the face, neck and trunk respond well to treatment during infancy. Complete obliteration of the stain is not possible. Most capillary malformations require multiple treatment sessions. Treatment can be initiated at any age. Surgical intervention may be required for associated soft tissue hypertrophy or limb length discrepancy



Lymphatic Malformations

Introduction
Lymphatic Malformation due to the failure of embryonic lymphatics or lymphatic jugular sacs to connect or drain into the venous system. Lymphatic malformations are quite variable in their presentation including
-
cystic lymphatic lesions
-
angiokeratoma
-
chylous leak conditions
-
osseous lesions of Gorham-Stout
-
generalized lymphatic anomaly
-
lymphedema
lymphatic malformations present as soft compressible masses. The overlying skin is normal. Occasionally, small fluid filled vesicles may be appreciated upon closer inspection of the overlying skin.
Lymphadema is classified into
-
primary (due to anomalous development) and
-
secondary (iatrogenic or due to obstruction and infection by Wucheria Bancroffti.
The protein rich fluid in the superficial interstitial space gets replaced by fibrotic tissue or adipose tissue.
Lymphatic malformations can also be classified into:
-
Microcystic <1cm in diameter, cannot be aspirated (common in oropharyngeal cavity): Lymphangioma Circumscriptum
-
Macrocystic >1cm in diameter, can be aspirated (common in neck and axillary region)
-
Combined Macro/Microcystic
-
Lymphangiomatosis (Pulmonary, pleural, intestinal)
-
Lymphadema
-
Mesenteric Cyst/Omental Cyst
What is the difference between an anterolateral neck lymphatic malfomation and a cystic hygroma?
Cystic hygroma is Most common left side posteriorly and can extend to oral cavity. Margins are ill defined, soft fluctuant, cystic, compressible but not reducible and brilliantly transilluminant.
Antenatal diagnosis of cystic hygroma:
Antenatal USG/MRI
Cystic lymph filled cavity in posterior or anteriolateral neck, increased nuchal translucency >2.5mm between 10th and 14th week (cystic hygroma), if the mass is cystic then it’s a LM, if not then teratoma
Antenatal Management
Risk of postnatal airway obstruction: → consider EXIT procedure (EXIT to airway management)
If solid then Definitive EXIT
Risk of Hydrops Fetalis secondary to large pleural effusion, consider antenatal intervention (amniotic shunting or repeated TAPS)
Treatment
Drainage: Needle aspiration done is not indicated for definitive treatment. Useful for emergency decompression of cyst to relieve airway obstruction to facilitate delivery
Surgical Excision: Preferred method of treatment. Downside is incomplete excision due to fluctuance of vital structures. Get CT or MRI to evaluate full extent of the disease. Traditional approach is an incision overlying the mass (neck scars are very unpleasing). Most recently some have used an endoscopic approach through anterior chest. If entire cyst is resected, recurrence is zero. Subtotal resection. (56%), partial resection (86%), drainage only (100%)
Sclerotherapy: OK432 (picibnil-an inactive strain of group A streptococcus pyogenes and has antineoplastic activity), Bleomycin, Doxycycline, Ethanol, Sodium Tetradecyl Sulfate (given at a dose of 1-3ml of 2% solution depening upon size of the lesion at an interval of 4 weeks)
Radiofrequency Ablation and laser excision: used for microcystic lymphatic malformation
Combined therapy: Combination of cyst aspiration and injection of OK 432
Antibiotic if infected
Investigations
Ultrasonography and contrast enhanced magnetic resonance imaging (MRI) are useful adjuncts to further characterize exact anatomic location and extent of lymphatic anomalies.
Treatment
-
The medical treatment of lymphatic malformations is varied and includes observation, pharmacotherapy, and sclerotherapy
-
Acute swelling of a lymphatic malformation near a vital structure such as the eye or airway can be managed with corticosteroids. Pain related to lymphatic malformations can be treated with analgesics and rest. An infected lymphatic malformation mandates antibiotic treatment.
-
Many pharmacotherapies have been proposed for lymphatic malformations. Sirolimus (rapamycin) has shown some early promise for patients with complex lymphatic disorders
-
Image guided percutaneous sclerotherapy is the often preferred primary treatment for lymphatic malformations. Sclerotherapy works especially well for macrocystic lesions and may render them undetectable. Sclerotherapy causes direct endothelial damage with subsequent luminal obliteration and fibrosis. Common sclerosants include pure ethanol, sodium tetradecyl sulfate and doxycycline. Doxycycline is the sclerosant of choice for macrocystic lymphatic malformations based on efficacy and safety [58]. Bleomycin is a reasonable option for microcystic disease especially of the head and neck because it causes less post-procedural swelling
-
For larger macrocysts with an aspiration volume greater than 30 mL, a small 6 or 8 French catheter may be placed percutaneously guided by ultrasound with a Seldinger technique. Aspiration and injection of the sclerosant may then be performed through the drainage catheter. Doxycycline is injected to at least 50% of the aspirated fluid volume under image guidance. The drain is then clamped for four hours of dwell time and access into the cyst is maintained. The drain is then unclamped and placed to gravity drainage. Additional sclerosant injections can then be performed over three to four days while the patient remains in the hospital. Key considerations include the need for general anesthesia, location of the lymphatic malformation, potential need for intensive care support post procedure, post-therapy swelling near the airway or in contained anatomical compartments and potential toxicities of the sclerosants. Edema and pain are expected consequences.
-
Sclerotherapy is associated with the need for multiple sessions, residual disease, residual cosmetic deformity, and potential toxicity of some of the sclerotic agents.
-
Lasers and radiofrequency ablative techniques may be used to treat lymphatic malformations. The carbon dioxide laser can be used to ablate mucosal lymphatic malformations. Patients report improvement in pain, bleeding and oral feeding [62][63]. Carbon dioxide laser is also used to treat lymphatic vesicles that grow through scars at sites of prior resections. Radiofrequency ablation can be utilized similarly
Surgical Therapy
The only potential curative therapy for lymphatic malformations is surgical resection. The goal is to ensure as complete of a resection as possible in the anatomic area. Repeat excision of the same area is technically challenging and increases morbidity. Dissection in areas previously treated with sclerotherapy is also challenging due to scarring and fibrosis.
The general principles regarding surgical resection for lymphatic malformations include
-
limiting the resection to a defined anatomic region
-
preserving neurovascular structures
-
minimizing blood loss and planning for replacement
-
performing closed suction drainage of the resection cavity
The recurrence rate after surgical resection is high due to the growth and expansion of abnormal lymphatics in the remaining, seemingly normal, yet involved tissue. Cutaneous warty lymphatic vesicles may develop in the scar. The scar can be revised if necessary. Lymphatic malformations do not exhibit enhancement with contrast except in cases of intralesional bleeding within the lymphatic cysts.
Surgical resection is associated with significant risks of hemorrhage, nerve damage, incomplete resection, residual lymphatic leaks, and recurrence.
RAPID RECALLS
-
Most frequent site of LM is neck and then axilla
-
They can occur anywhere except the brain
-
Positive for podoplanin and LYVE I
-
Associated with downs syndrome and turners syndrome and Gorham Stout Syndrome
-
Associated with cardiac malformation and Aneuploidy
-
Clinical features are: Proptosis (Periorbital/orbital lesions), Facial LMs (Associated with skeletal overgrowth, macroglossia), Airway obstruction (tongue)
-
Pleural/Pericardial effusion
-
GI LMs 🡪 protein losing enteropathy
-
Pelvic can cause urinary and fecal incontinence
-
LMs in extremity can cause bone resorption called Gorham Stout Syndrome 🡪 Vanishing bone disease
-
Diagnosis by USG, CT/MRI, Lymphangiography, Lymphoscintography
-
Possible complications include: infection, bleeding, airway obstruction, respiratory distress.
-
Treatment options include: sclerotherapy, surgical resection
-
Sclerosants include: pure ethanol (70%), sodium tetracycline sulfate, doxycycline, bleomycin (0.3mg/kg – 0.6mg/kg) every 6 weeks. Bleomycin injection is 15mg and is diluted in in distal water. After aspiration of as much fluid as possible inject using the same needle. Observe for 2 hours for anaphylactic reaction.
-
Sirolimus 0.08mg/m2 BD for 3 months [1mg tablet], with dose adjustment (6mg/d) based on serum levels of sirolimus (target 4-12ng/ml) at day 15, then almost once a month. S/E: leucopenia. It works by blocking mamillian target of rapamycin (mTOR) serine, threonine kinase receptor regulated by phosphoinositol 3 kinase. Blocks angiogenesis as well as T cell proliferation.
-
Differential Diagnosis can include a cystic teratoma, in that case ultrasound will reveal a solid component or calcifications. Then perform cross sectional imaging by a CT scan or MRI. Chest X ray to rule out mediastinal malformation and a serum AFP level.
-
Ultrasound can give vital information about its location, septations, involvement of the carotid artery, internal jugular vein or trachea.
-
There is increasing evidence in the literature that more than one-third of asymptomatic cervical lymphatic malformations may regress significantly or resolve completely with no intervention. Anecdotally, cervical malformations present at birth may be the most likely to resolve, as opposed to those that appear later in infancy or childhood.
-
A lesion volume ratio (LVR) has been described to objectively assess lymphatic malformation in utero. This is obtained by calculating the volume of the lesion using the ellipsoid formula (length × height × width × 0.52) and dividing it by the head circumference.
-
When found in the soft tissues, macrocystic lesions typically have a soft, fluctuant, ballotable consistency. Microcystic or mixed lesions may have a very firm consistency, giving the impression of a solid mass. The overlying skin may not be involved or may contain vascular lesions
-
Lymphatic malformations present the surgeon with a dilemma. They are benign lesions that do not respect tissue planes and often demonstrate an infiltrative effect that involves vital neurovascular structures. Complete excision therefore may result in severe morbidity, whereas incomplete excision is associated with a high risk of residual lesion or recurrence. In addition, resection of large lesions may result in significant blood loss.
-
While resecting the cervical component, take care of teh brachial plexus and while resecting the axillary section, take care of the thoracic and thoracodosorsal nerves
-
The walls were lined by flattened endothelium and the cysts appeared empty or contained proteinaceous material. The endothelium stains positive for D2-40. This is an immunohistochemical marker that reacts with a sialoglycoprotein present on the surface of lymphatic endothelium. It helps differentiate between arteriovenous lesions and lymphatic malformations.




Venous Malformations
Venous Malformation are slow flow lesions. Venous malformations arise due to a developmental defect of the vasculature. most commonly involve the skin and soft tissues but can be located anywhere in the body. Some venous malformations exhibit expansion when placed in a dependent position or with a Valsalva maneuver. Superficial venous malformations of the skin and soft tissues often present a soft, bluish, easily compressible mass. Venous malformations grow commensurate with the growth of the child. Venous malformations tend to expand during adolescence suggesting that pubertal hormones may contribute to their expansion.
Investigations
Ultrasonography, MRI and venography are useful imaging modalities to assess venous malformations. Doppler ultrasonography reveals a soft, compressible mass with monophasic low flow velocity. MRI depicts the relationship of the venous malformation to other structures and its size. Lesions are hyperintense with T2 sequences. Septations and venous spaces can be observed. Venous malformations enhance with the administration of contrast. Lymphatic malformations do not exhibit enhancement with contrast except in cases of intralesional bleeding within the lymphatic cysts.
Gastrointestinal venous malformations may be solitary or multifocal. The lesions may be small or massive and can involve any or all of the intestinal wall. The majority of focal gastrointestinal venous malformations occur as transmural lesions of the left colon and rectum. Flow in the marginal venous system is sluggish, predisposing the patient to thromboembolism and thrombophlebitis. Anomalous connections of the marginal venous system to the iliac vein and inferior vena cava is a risk factor for pulmonary embolism.
Treatment
Medical interventions include compression, anti-inflammatory medications and sclerotherapy. Consideration should be given to treatment with low molecular weight heparin to improve the coagulation profile for patients with elevated D-dimer levels during the perioperative period, pregnancy, bed rest or travel. Sclerotherapy can be effective for symptomatic venous malformations. Sclerosants include dehydrated ethanol and sodium tetradecyl sulfate. Large venous malformations are accessed by direct puncture under ultrasonography and fluoroscopy. Compression and tourniquets limit venous drainage and systemic delivery of the sclerosant. Large draining veins may require embolization techniques with coils or liquid embolics to augment the effects of sclerotherapy.
Surgery
Surgical resection of venous malformations is ideal for focal lesions. Preoperative sclerotherapy may allow resection of larger lesions. Staged subtotal resections may be necessary. Patients with large venous malformations should be evaluated for a localized intravascular coagulopathy prior to surgical intervention. Diffuse colorectal venous malformations may be treated with colectomy, anorectal mucosectomy and coloanal pull through


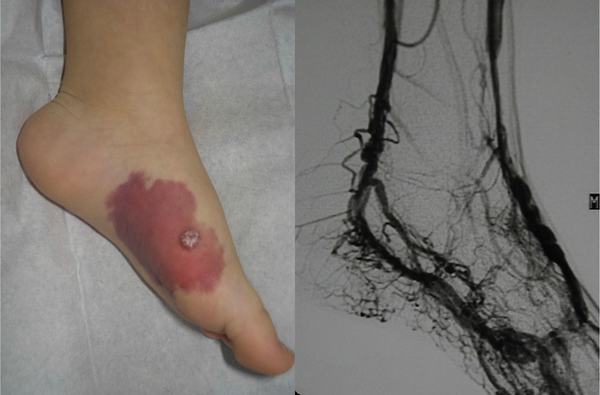
Arteriovenous Malformations
Introduction
Arteriovenous Malformation The nidus, or central core, is composed of abnormally connected arteries and veins that form shunts. These shunts bypass the high resistance capillary bed. During childhood and adolescence they become more obvious due to the presence of a palpable thrill under the warm, pink stain. Manual compression of feeding arteries and draining veins results in a slowing of the heart rate known as Nicoladoni-Branham sign. As they expand, they become more mass like. The skin and overlying soft tissue can bleed or ulcerate. Heart failure is a late consequence.
Pain, ulceration and heart failure are some of the serious side effects of arteriovenous malformations. Pain and ulceration results from pure arterial ischemia due to proximal steal through the malformation. Additionally, venous hypertension alters the capillaries reducing the diffusion of oxygen and nutrients from the capillary bed to the soft tissues.
Arteriovenous malformations tend to progress with time and can be documented by a clinical staging system
Investigations
Ultrasonography with color and spectral Doppler evaluation can reliably detect arteriovenous shunting. Radiographically, there is not a discrete parenchymal mass. MRI allows for assessment of the adjacent soft tissue. MRA can depict dilated feeding and draining veins. The nidus may or may not be visible. Edema, increased fat, muscle enlargement and bony changes are variably present.
Treatment
The goal of treatment is to control shunting and palliate symptoms. The majority of arteriovenous malformations require treatment due to progressive expansion. The mainstays of therapy include angiographic embolization, surgical resection or a combination of the two. Regardless of the method of treatment, proximal feeding arteries must never be ligated or embolized. Ligation may temporarily relieve heart failure, pain, or bleeding but prevents future embolizations. Despite ligation, the nidus of the arteriovenous malformation will recruit nearby arteries and veins resulting in continued growth and expansion Combined approaches utilizing embolization first and then surgery two to three days later is a reasonable approach for well localized arteriovenous malformations. Preoperative embolization diminishes bleeding and may define the resection margin
Surgery
The surgical goal is to completely resect the nidus and overlying soft tissue and skin thus minimizing the risk of recurrence. The planned resection is determined by studying the earliest radiologic imaging prior to any interventions.